Now, as to whether or not Malone is a liar, which is Jack's (second-hand) contention. (Second-hand because Jack presumably lacks the credentials to know, himself, whether or not Malone's telling the truth. He's getting that position from Twitter, CNNLOL, et al.):
What does Malone get by lying? Is somebody bankrolling him to fabricate these patents and papers:
A novel approach to study packaging of retroviral RNA by RNA transfection (Abstract). RW Malone, P. Felgner, I. Verma. RNA Tumor Viruses, May 17-18, 1988. Cold Spring Harbor
mRNA Transfection of cultured eukaryotic cells and embryos using cationic liposomes. Malone RW. Focus. 1989; 11:61-8
DNA and RNA Transfection and Vaccination (Abstract). First Place, Northwestern AOA Research Symposium Competition for Medical Students: 1989.
Cationic liposome-mediated RNA transfection. Malone RW, Felgner PL, Verma IM. Proc Natl Acad Sci (PNAS) U S A. 1989;86(16):6077-81. Cited in 749 articles.
Direct gene transfer into mouse muscle in vivo. Wolff JA, Malone RW, et al. Science. 1990;247(4949 Pt 1):1465-8. Cited in 4,750 articles.
High levels of messenger RNA expression following cationic liposome mediated transfection tissue culture cells. Malone R, Kumar R, Felgner P. NIH Conference: “Self-Cleaving RNA as an Anti-HIV Agent (abstract). Washington, DC June 1989.
Cationic liposome-mediated RNA transfection. Dwarki VJ, Malone RW, Verma IM. Methods Enzymol. 1993;217:644-54. Cited in: 102 articles.
Delivery of exogenous DNA (includes mRNA) sequences in a mammal P Felgner, JA Wolff, GH Rhodes, R Malone, D Carson. Biotechnology Advances 1993: 15 (3-4), 763-763
Lipid-mediated polynucleotide administration to deliver a biologically active peptide and to induce a cellular immune response (includes mRNA). Assigned to Vical, Inc and licensed to Merck. No. 7,250,404, date of issue: 7/31/07 Cited in 105 articles. Priority Date: 3/21/1989.
Lipid-mediated polynucleotide administration to reduce likelihood of subject’s becoming infected (includes mRNA). Assigned to Vical, Inc and licensed to Merck. US Pat. Ser. No. 6,867,195 B1. Date of issue: 3/15/05. Priority Date: 3/21/1989.
Generation of an immune response to a pathogen (includes mRNA). Assigned to Vical, Inc and licensed to Merck. US Pat. Ser. No. 6,710,035. Date of issue: 3/23/04. Citations: 39 articles. Priority Date: 3/21/1989.
DNA (and mRNA) vaccines for eliciting a mucosal immune response. US Pat. Ser. No. 6,110,898, date of issue: 8/29/00. Cited in 40 articles.
Expression of exogenous polynucleotide sequences in a vertebrate, mammal, fish, bird or human (includes mRNA) . Assigned to Vical, Inc, licensed to Merck. US Pat. Ser. No. 6,673,776. Date of issue: 1/6/04. Priority Date: 3/21/1989.
Methods of delivering a physiologically active polypeptide to a mammal (includes mRNA). Assigned to Vical, Inc, licensed to Merck. US Pat. Ser. No. 6.413.942. Date of issue: 7/2/02. (cited in 150 articles). Priority Date: 3/21/1989.
Induction of a protective immune response in a mammal by injecting a DNA sequence (includes mRNA). Assigned to Vical, licensed to Merck. US Pat. Ser. No. 6,214,804, date of issue: 4/10/01. Cited in 360 articles. Priority Date: 3/21/1989.
DNA vaccines for eliciting a mucosal immune response (includes mRNA). US Pat. Ser. No. 6,110,898. Inventors: RW Malone and Jill Glasspool Malone. Date of issue: 8/29/00. Cited in 40 articles. Priority Date: 1997.
Induction of a protective immune response in a mammal by injecting a DNA sequence (includes mRNA). Assigned to Vical, Inc, licensed to Merck. US Pat. Ser. No. 5,589,466. Date of issue: 12/31/96. Cited in 899 articles. Priority Date: 3/21/1989.
Delivery of exogenous DNA sequences in a mammal (includes mRNA). Assigned to Vical, Inc, licensed to Merck. US Pat. Ser. No. 5,580,859. Date of issue: 12/3/96. Cited in 1244 articles. Priority Date: 3/21/1989.
Generation of antibodies through lipid mediated DNA delivery (includes mRNA). Assigned to Vical, Inc, licensed to Merck. US Pat. Ser. No. 5,703,055. Date of issue: 12/30/97. Cited in 419 articles. Priority Date: 3/21/1989.
Cationic liposome-mediated RNA transfection. Dwarki VJ, Malone RW, Verma IM. Methods Enzymol. 1993;217:644-54. Cited in: 88 articles.
Robert Malone’s patents issued cationic lipid formations for use in mRNA vaccinations
Formulations and methods for generating active cytofectin: polynucleotide transfection complexes. US Pat. Ser. No. 5,925,623 7/20/99.
Cationic Transport Reagents. US Pat. Ser. No. 5,892,071 issued 4/06/99.
Polyfunctional cationic cytofectins, formulations and methods for generating active cytofectin: polynucleotide transfection complexes. US Pat. Ser. No. 5,824,812 issued 10/20/98.
Cationic Transport Reagents. US Pat. Ser. No. 5,744,625 issued 4/28/98.
Cationic Transport Reagents. US Pat. Ser. No. 5,527,928, date of issue: 6/18/96.
Papers related to cationic lipid polynucleotide transfection and vaccination (including mRNA)
Electroporation enhances transfection efficiency in murine cutaneous wounds. Byrnes CK, Malone RW, et al. Wound Repair Regen. 2004;12(4):397-403.
Marked enhancement of macaque respiratory tissue transfection by aurintricarboxylic acid. Glasspool-Malone J, …, Malone RW. Gene Med. 2002;4(3):323-2.
Enhancing direct in vivo transfection with nuclease inhibitors and pulsed electrical fields. Glasspool-Malone J, Malone RW. In Gene Therapy Methods: Methods Enzymol. 2002;346:72-91
Cutaneous transfection and immune responses to intradermal nucleic acid vaccination are significantly enhanced by in vivo electropermeabilization. Drabick JJ, Glasspool-Malone J, …, Malone RW. Mol Ther. 2001;3(2):249-55. Cited in 192 articles.
Theory and in vivo application of electroporative gene delivery. Somiari S, Glasspool-Malone J, … Malone RW. Mol Ther. 2000;2(3):178-87. Cited in 345 articles.
Efficient nonviral cutaneous transfection. Glasspool-Malone J, …, Malone RW. Mol Ther. 2000;2(2):140-6. Cited in 138 articles.
Developing dendritic cell polynucleotide vaccination for prostate cancer immunotherapy. Berlyn KA, …, Malone RW J Biotechnol. 1999;73(2-3):155-79
Models of Cationic Liposome Mediated Transfection. Gene Therapy and Molecular Biology. Ahearn A, Malone RW. Vol 4. Gene Therapy and Molecular Biology 1999;4
Cationic lipid-mediated gene delivery to murine lung: correlation of lipid hydration with in vivo transfection activity. Bennett MJ, …, Malone RW, Nantz MH. J Med Chem. 1997;40(25):4069-78
Toxicity of cationic lipid-ribozyme complexes in human prostate tumor cells can mimic ribozyme activity. Freedland SJ, Malone RW, et al. Biochem Mol Med. 1996;59(2):144-53
Considerations for the design of improved cationic amphiphile-based transfection reagents. Bennett MJ, …, Malone RW. Journal of Liposome Research 1996;6(3):545-65
Structural and functional analysis of cationic transfection lipids: the hydrophobic domain. Balasubramaniam RP, …, Malone RW. Gene Ther. 1996;3(2):163-72. cited in 172 articles.
Direct gene tranfer into mouse muscle in vivo. N Shafee, …, RW Malone, et al. International Journal of Virology 2 (1), 33-38
A flexible approach to synthetic lipid ammonium salts for polynucleotide transfection. MJ Bennett, RW Malone, MH Nantz. Tetrahedron letters 36 (13), 2207-2210
Tfx-50 Reagent, a new transfection reagent for eukaryotic cells. Schenborn E, …, Malone RW, et al. 1995
So, Jack, if Malone is lying, what's he getting out of it, other than a long and well documented history of expertise on the subject matter he's lying about?
Now, then: If he's right about mass formation psychosis, I should expect attacks instead of a clear-eyed rebuttal.
Of course, I'd also expect Google to hastily scrub 'mass formation psychosis' from search results.
Oh, shit, they did.
Ministry of Truth tactic should tell you all you need to know about what really motivates Google not just censoring videos or people, but now even concepts and theories in real time by Jamie White Dr. Robert Malone, the virologist who invented the mRNA technology used in the COVID injection...
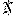
newsrescue.com
When it comes to figuring out who's lying, follow the money. Malone isn't getting paid, he's getting censored.